Introduction
Despite advancements in cancer treatments, the global incidence of cancer deaths remains a significant concern. While medical progress extends the lives of patients with metastatic disease, this longevity underscores the increasing demand for comprehensive supportive and palliative care (PC). International medical societies advocate for the early integration of palliative care, ideally alongside specific cancer treatments, to improve patient outcomes and quality of life. Research consistently demonstrates that early palliative care intervention reduces symptom burden, alleviates depressive symptoms, and enhances overall quality of life for patients. Furthermore, studies have indicated that concurrent medical oncology and palliative care approaches can prolong survival and improve the management of advanced-stage patients nearing end-of-life.
Integrating palliative care within comprehensive cancer centers is particularly crucial. However, the current capacity of palliative care teams often struggles to meet the escalating needs. To optimize patient care, oncology teams require effective methods to identify patients who would most benefit from timely palliative care referral. While prognostic scores exist, their practical application in daily clinical decision-making is limited. These tools, often designed for hospice or end-of-life settings, primarily predict short-term mortality rather than pinpointing the broader need for palliative care intervention. Consequently, healthcare professionals often find prognosis-based criteria too restrictive and less helpful than need-based approaches for initiating palliative care.
Recognizing these challenges, the French society for palliative support and care (SFAP) developed the PALLIA-10 questionnaire in 2010. This 10-item screening tool is designed for use by any caregiver to assess medical, psychosocial, and ethical dimensions of patient needs. The PALLIA-10 assigns a score from 0 to 10, aiming to identify patients who require palliative care services. Current recommendations suggest referring patients with a PALLIA-10 score exceeding 3 to specialized palliative care teams. However, given that comprehensive cancer centers typically manage patients with advanced and heavily treated cancers, there is a need to refine the referral criteria to ensure appropriate and timely palliative care intervention. This study investigates the application of the PALLIA-10 questionnaire in hospitalized advanced cancer patients within French comprehensive cancer centers, specifically exploring the suitability of the score threshold of 3 for palliative care referrals.
Methods
2.1. Patient Population and Study Design
This prospective, multicenter study was conducted across 18 French comprehensive cancer centers. The study population included adult patients hospitalized in conventional medicine or radiotherapy departments. Patients in surgical units and outpatients were excluded from this study. All participants, and their family caregivers where applicable, were provided with detailed information about the study and had the option to decline participation. The study was approved by relevant French ethics committees (CCTIRS and CNIL), registered with ClinicalTrials.gov (NCT02479061), and adhered to ethical guidelines for research involving human subjects.
Data collected included patient demographics (age, gender, family status, and Karnofsky performance score, converted to ECOG-PS), disease characteristics (diagnosis, stage), and aspects of medical care management (reason for hospitalization, palliative care needs). The most recent biological data available within three weeks of enrollment were also recorded.
The PALLIA-10 questionnaire was administered to patients with incurable cancer, based on the treating physician’s assessment. A national initiation meeting was held to standardize procedures for enrollment, questionnaire administration, and data collection across all participating centers. Each participating team, comprising at least one physician and one nurse from a supportive care unit, received specific training on utilizing the PALLIA-10 questionnaire and scoring according to SFAP guidelines. The SFAP guidelines emphasize that any healthcare professional, regardless of their background, should be able to complete the questionnaire. Patients or their caregivers were directly interviewed. Data collection occurred on a single designated day at each institution to ensure a uniform, point-prevalence assessment. Disease staging was classified using the Krakowski classification system to determine the palliative care phase (curative, early palliative, terminal palliative, or agonic).
Follow-up data, including survival status at 6 months and the date of palliative care initiation for patients not receiving it at study enrollment, were collected.
2.2. Study Outcomes
The primary outcome was to determine the prevalence of palliative patients with a PALLIA-10 score greater than 3 among hospitalized cancer patients in French cancer centers. Secondary outcomes included: the proportion of patients already receiving palliative care at the time of the study or referred within six months, the prevalence of patients with a PALLIA-10 score exceeding 5, and overall survival (OS) based on the PALLIA-10 score thresholds of 3 and 5.
2.3. Statistical Analysis
The analysis focused on the palliative patient cohort. Palliative status was determined by a documented palliative entry date or, in its absence, by the medical oncologist’s assessment based on the Krakowski classification. Descriptive statistics summarized baseline patient characteristics. Overall survival was calculated from the date of study inclusion to the date of death or last follow-up, censored at 6 months. Kaplan-Meier survival curves and log-rank tests were used to compare survival across PALLIA-10 score groups.
Cox proportional hazards models were used to assess the association of PALLIA-10 scores (≤3, 4-5, and >5) with overall survival, adjusting for potential confounding variables. These variables included age, reason for hospitalization, tumor type, palliative care management at inclusion, agreement among healthcare teams regarding palliative care needs, Karnofsky score, number of metastatic sites, and biological markers (hemoglobin, lymphocytes, LDH, albumin, and CRP). Variable selection for multivariate models was performed using a backward selection procedure to retain factors significant at the 5% level.
Logistic regression analysis was employed to evaluate factors associated with palliative care referral. Initially, the analysis examined PALLIA-10 score as a binary variable (≤3 vs. >3), and subsequently as a categorical variable (≤3, 4-5, and >5). Other factors considered were family status, age, reason for hospitalization, tumor type, opinion convergence, Karnofsky score, number of metastatic sites, and biological markers. Variable selection for multivariate logistic regression also used a backward selection procedure to maintain variables significant at the 5% level.
Receiver operating characteristic (ROC) curve analysis was conducted to assess the sensitivity and specificity of PALLIA-10 scores in predicting palliative care needs. The Youden index J was calculated to identify the optimal PALLIA-10 score cut-off point. A p-value <0.05 was considered statistically significant for all analyses.
Results
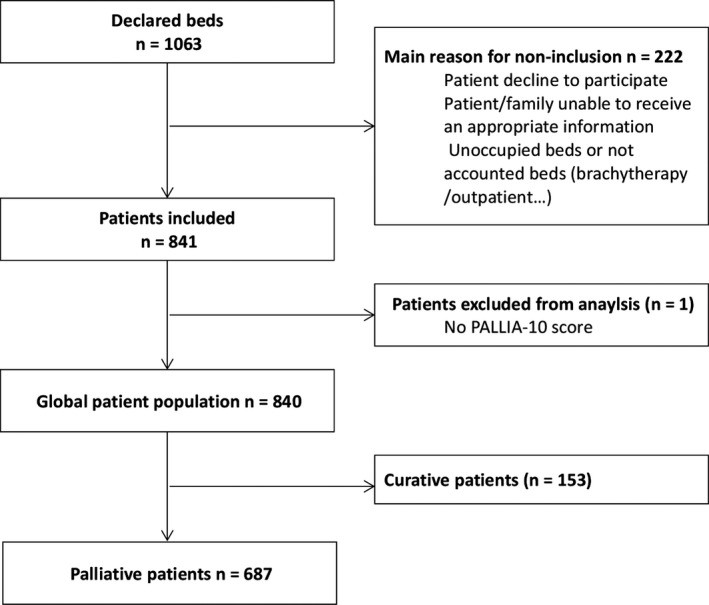
The study involved 18 French Comprehensive Cancer Centers (90% of the total centers). Data collection occurred on a single day in each of sixteen centers during June 2015, and in two additional sites in October 2015. These centers collectively reported 1063 hospital beds. After applying exclusion criteria (minors, outpatients, brachytherapy, unoccupied beds), 841 patients were assessed, of whom 687 (82%) were classified as palliative care patients (Figure 1). The PALLIA-10 score was missing for one patient.
Figure 1. Trial profile. Reasons for non-inclusion have not been quantified.
The median age of participants was 64.8 years (range 19-92), and 54% were female. The majority (71.3%) lived with a caregiver. A significant proportion (85.4%) had a poor performance status (ECOG-PS ≥ 2). The most common primary cancer sites were digestive tract (18.3%), breast (17.6%), and lung/pleura (14.7%). Metastatic disease was present in 87.4% of patients, with 69.3% having more than two metastatic sites. Reasons for hospitalization included treatment administration (30.9%), acute complications (20.0%), and symptom management (49.2%). The median time from hospitalization to study inclusion was 6 days. Most patients (97.2%) had a palliative care entry date, with a median interval of 3 months between initial diagnosis and palliative setting. At study inclusion, 31.4% were already receiving palliative care, with a median of 20.7 months from diagnosis to palliative care initiation. Reasons for palliative care requests were predominantly symptom management (74.5%) and psychological support (40.3%). Patient demographics and baseline characteristics are detailed in Table 1.
Table 1. Main demographics and baseline characteristics. Data are median (range min-max) or n (%)
| Patients in palliative setting N = 687 | |
|---|---|
| Median age at inclusion (years) | 64.8 (19–92) |
| Median age at diagnosis (years) | 61.6 (12–90) |
| Gender | |
| Female | 371 (54%) |
| Male | 316 (46%) |
| Familial status | |
| Missing data | 1 |
| Alone | 167 (24.3%) |
| Caregiver at homea | 490a (71.4%) |
| Dependent person at home | 29 (4.2%) |
| Performance Status (ECOG-PS) | |
| 0 | 5 (0.7%) |
| 1 | 95 (13.8%) |
| 2 | 202 (29.4%) |
| 3 | 210 (30.6%) |
| 4 | 175 (25.5%) |
| Main primary tumor localizationsa | |
| Digestive tract | 126 (18.3%) |
| Breast | 121 (17.6%) |
| Lung and/or pleura | 101 (14.7%) |
| Head and Neck | 63 (9.2%) |
| Urologic | 82 (11.9%) |
| Gynaecologic | 78 (11.4%) |
| Metastatic disease | |
| Missing data | 2 |
| Metastatic disease | 599 (87.4%) |
| Median number of metastatic sites | 2.0 (0–8) |
| Reason(s) for hospitalization | |
| Treatment (chemotherapy, radiotherapy…) or health assessments | 212 (30.9%) |
| Acute complication (aplasia, sepsis, Intracranial hypertension…) | 137 (20.0%) |
| Symptoms (pain, dyspnea…) | 338 (49.2%) |
| Median delay between hospitalization date and inclusion (days) | 6.0 (0–77) |
| Patients with entry date in palliative setting | 668 (97.2%) |
| Median delay between initial diagnosis and palliative settinga (months) | 3.0 (0–414) |
| Palliative care management initiated at the time of the inclusion | 216 (31.4%) |
| Median delay between initial diagnosis and palliative care initiation (months) | 20.7 (0–374) |
| Reasons for palliative care request | |
| Symptoms | 161 (74.5%) |
| Psychological support | 87 (40.3%) |
| Orientation of the patient | 77 (35.6%) |
| Support for the patient’s family | 40 (18.5%) |
| Healthcare team support | 24 (11.1%) |
| Terminal accompaniment | 18 (8.3%) |
17 (2.5%) patients have a caregiver and one dependent person at home.
aTumor localizations accounting for at least 10% of the patients. ECOG-PS: Eastern Cooperative Oncology Group Performance Status.
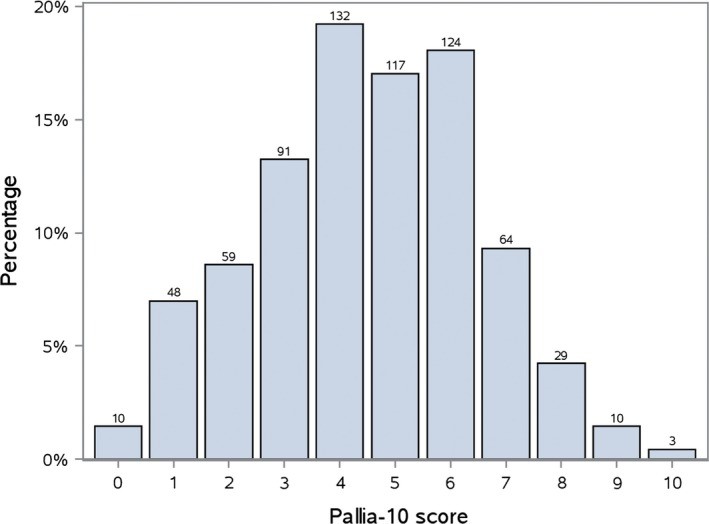
The distribution of PALLIA-10 scores is shown in Figure 2. The prevalence of palliative patients with a PALLIA-10 score greater than 3 was 69.7% (95%CI 66.1%-73.1%), and for a score greater than 5, the prevalence was 33.5% (95%CI 30.0%-37.1%).
Figure 2. Distribution of PALLIA-10 scores in the palliative population (n = 687). Ten palliative patients had a PALLIA-10 score of 0.
3.1. Palliative Care Management
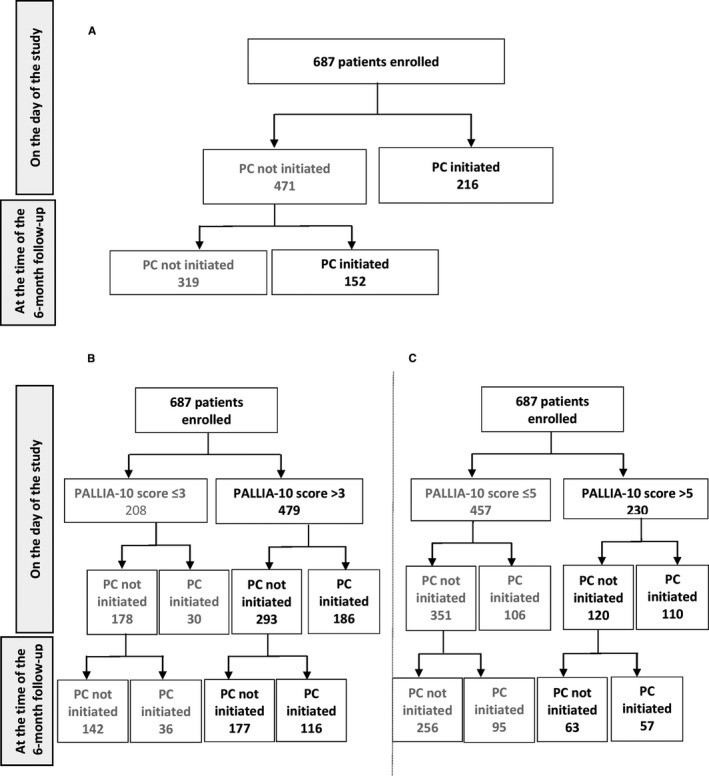
At the time of the study, 31.4% of patients (n=216), with a median PALLIA-10 score of 6 (range 0-9), were already receiving palliative care. Among patients with PALLIA-10 scores >3, 38.8% were receiving palliative care. This proportion increased to 47.8% for patients with scores >5. The 69% of patients (n=471) not receiving palliative care had a median PALLIA-10 score of 4 (range 0-10). Within this group, 61.2% of patients with PALLIA-10 scores >3 and 52.2% with scores >5 were not receiving palliative care (Figure 3).
Figure 3. Distribution of palliative care intervention in the palliative patient population at study enrollment and 6 months post-enrollment (A), and distribution of the palliative patient population by PALLIA 10 score ((B) score >3 and (C) score >5) and palliative care intervention at study enrollment and 6 months post-enrollment.
Within six months following the study, 22.1% (n=152) of patients initiated palliative care. These patients had a median PALLIA-10 score of 4.5 (range 0-10) at study enrollment. Among them, 24.2% had a PALLIA-10 score >3 and 24.8% had a score >5.
Logistic regression analysis showed that a PALLIA-10 score >3 was significantly associated with palliative care referral (adjusted OR 2.6, 95%CI 1.65-4.11). Further analysis revealed adjusted ORs of 1.9 (95%CI 1.17-3.16) for PALLIA-10 scores of 4-5 and 3.59 (95%CI 2.18-5.91) for scores >5, indicating increasing likelihood of palliative care referral with higher PALLIA-10 scores. This association remained significant after adjusting for factors like metastatic sites, agreement among healthcare teams, and reasons for hospitalization (Table 2).
Table 2. Predictive factors for palliative care team intervention. Final multivariate model of predictive factors of palliative care management
| Variables | OR | 95%CI | P-value |
|---|---|---|---|
| PALLIA 10 Score | [0–3] (Ref.) | 1.00 | |
| [3–5] | 1.924 | [1.169–3.165] | |
| [5–+] | 3.595 | [2.185–5.914] | |
| Number of metastatic sites | No metastatic site (Ref.) | 1.00 | 0.0294 |
| One metastatic site | 0.426 | [0.214–0.846] | |
| At least two metastatic sites | 0.663 | [0.356–1.235] | |
| Opinion convergence (oncologist/health team/Palliative care team) | At least one disagree (Ref.) | 1.00 | 0.0002 |
| All agree | 3.942 | [1.939–8.017] | |
| Reasons of hospitalisation | Treatment (Ref.) | 1.00 | |
| Complications | 2.731 | [1.562–4.777] | |
| Symptoms | 3.132 | [1.949–5.033] |
Predefined potential predictive factors were familial status, age at inclusion, reason for hospitalization, type of tumor, the opinion convergence between oncologist, healthcare team and palliative care team, Karnofsky score, number of metastatic sites, PALLIA 10 score (0–3, 4–5, 5–+), and biological parameters (hemoglobin, lymphocytes, LDH, albumin, and CRP). Variables significant at a 20% level with less than 10% missing values in univariate analysis were used in a backward selection procedure to keep factors significant at a 5% level in the final predictive multivariate model.
ROC analysis identified a PALLIA-10 score of 5 as the optimal cut-off point for sensitivity and specificity in predicting palliative care needs (Supplementary materials p4 of original article).
3.2. Overall Survival (OS)
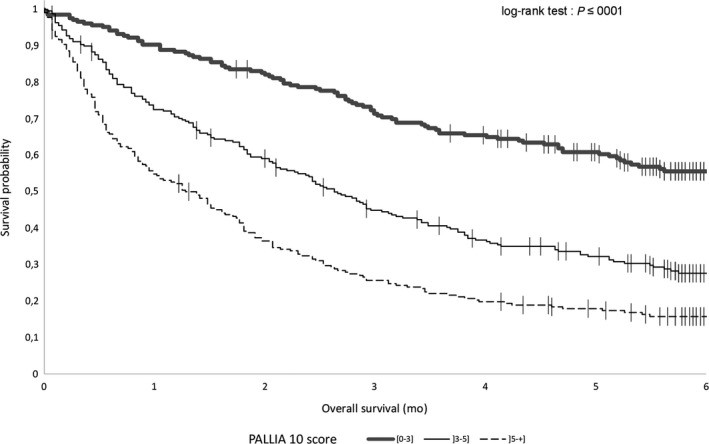
The median overall survival for palliative patients was 2.73 months (95%CI 2.43-3.06). Median OS varied significantly with PALLIA-10 scores (Figure 4). Median OS was not reached for patients with scores ≤3, 2.6 months (95%CI 2.1-3.2) for scores 4-5, and 1.3 months (95%CI 0.95-1.7) for scores >5.
Figure 4. Overall survival. Kaplan-Meier survival curves for patients with PALLIA-10 score ≤3, 3-5, and >5 in the 687 palliative patients.
PALLIA-10 score was a significant prognostic factor for survival, even after adjusting for Karnofsky index, reasons for hospitalization, tumor type, opinion convergence, metastatic sites, and lymphocyte levels. Compared to scores ≤3, the adjusted hazard ratios (HR) were 1.58 (95%CI 1.20-2.08) for scores 4-5 and 2.18 (95%CI 1.63-2.92) for scores >5 (Table 3). When considering PALLIA-10 as a continuous variable, a significant gradient was observed (adjusted HR 1.18, 95%CI 1.11-1.24), indicating a decrease in overall survival with each unit increase in PALLIA-10 score.
Table 3. Prognostic factors for OS. Final model of the multivariate Cox analysis of predefined potential prognostic factors (age at inclusion, reason for hospitalization, type of tumors, palliative care management at the time of inclusion, the convergence of opinions between the oncologist, the healthcare team and the palliative care team, Karnofsky score, number of metastatic sites, range of PALLIA 10 score (0–3, 4–5, and 5–+), and biological parameters (hemoglobin, lymphocytes, LDH, albumin, and CRP) on OS
| Variables | Hazard Ratio | 95%CI | P-value |
|---|---|---|---|
| PALLIA 10 Score | [0–3] (Ref.) | ||
| ]3–5] | 1.582 | [1.203–2.082] | |
| ]5–+] | 2.181 | [1.628–2.923] | |
| Karnofsky Score | >50 (Ref.) | ||
| ≤50 | 2.084 | [1.667–2.606] | |
| Reasons of hospitalisation | Treatment (Ref.) | 0.0002 | |
| Complications | 1.091 | [0.806–1.478] | |
| Symptoms | 1.597 | [1.248–2.044] | |
| Type of tumour | Breast (Ref.) | ||
| Head and neck | 1.747 | [1.140–2.678] | |
| Bone and soft tissue | 1.016 | [0.641–1.609] | |
| Lung and pleural | 1.939 | [1.403–2.682] | |
| Digestive tract | 1.662 | [1.212–2.279] | |
| Gynecologic | 0.894 | [0.610–1.310] | |
| Urologic | 1.100 | [0.765–1.582] | |
| Others | 0.908 | [0.585–1.410] | |
| Opinion convergence (oncologist/health team/Palliative care team | At least one disagree (Ref.) | 0.0019 | |
| All agree | 1.672 | [1.209–2.312] | |
| Number of metastatic sites | No metastatic site (Ref.) | 0.0003 | |
| One metastatic site | 0.802 | [0.535–1.200] | |
| At least two metastatic sites | 1.311 | [0.886–1.940] | |
| Lymphocytes | >0.7 G/l (Ref.) | 0.0047 | |
| ≤0.7 G/l | 1.337 | [1.093–1.636] |
Factors selected for the multivariate analysis (i.e., variables significant at a 20% level with less than 10% missing values in univariate analyses were included in a backward selection procedure to keep factors significant at a 5% level in the final Cox prognostic multivariate model).
Discussion
This PREPA-10 study is the first prospective, multicenter investigation to evaluate the PALLIA-10 questionnaire as a Screening Tool For Palliative Care Referrals in hospitalized patients. Our findings demonstrate that PALLIA-10 is a user-friendly and valuable instrument for identifying cancer inpatients in comprehensive cancer centers who are in need of palliative care. The questionnaire was easily integrated into standard hospital procedures, facilitating efficient communication among healthcare teams. The rapid scoring process was consistently achievable for almost all patients. The broad participation of 18 French cancer centers enhances the generalizability of our results. Healthcare teams across participating centers welcomed the questionnaire, and its implementation by various caregivers was successful across all sites.
The lack of a universal definition for the palliative care setting complicates the implementation of palliative care guidelines. Relying solely on a medical oncologist’s opinion to determine palliative status can lead to inaccurate and overly optimistic survival predictions. Prognostic tools can aid decision-making, but their utility is often limited. While the NCCN Clinical Practice Guidelines in Oncology for Palliative Care exist, their two-step screening process may be too sensitive for routine use. The PALLIA-10 questionnaire, recommended by SFAP in France since 2010, offers a practical, one-step approach to identify unmet palliative care needs. However, despite the recognized need for decision support tools for palliative care initiation, and the availability of PALLIA-10, its routine use has been limited due to a lack of robust evidence. PALLIA-10’s advantage lies in its multidimensional approach, addressing clinical, biological, psychosocial, and ethical aspects, which is crucial when self-assessment can be challenging. This comprehensive assessment, involving input from various healthcare professionals, strengthens the decision-making process for palliative care referral.
The strength of PALLIA-10 is its holistic approach, incorporating not only clinical and biological factors but also psychosocial, sociocultural, and ethical considerations. This aligns with the core principles of palliative care. Our study provides statistical evidence supporting the robustness of this approach. We advocate for the wider adoption of PALLIA-10 as a simple, multidimensional tool for palliative care screening. While this study did not aim for psychometric validation, the predictive value of the PALLIA-10 score for palliative care referral is evident. Further qualitative research is warranted to explore the individual impact of each PALLIA-10 item, particularly the innovative psychosocial criteria, to optimize the questionnaire’s use.
Our results indicate that while both PALLIA-10 score thresholds of >3 and >5 are relevant, a score >3, as currently recommended, might lead to over-referral in comprehensive cancer centers, given the current capacity of palliative care teams. Our ROC analysis suggests that a threshold of >5 may be more appropriate in this setting. We observed that patients with higher PALLIA-10 scores were more likely to receive palliative care, even after adjusting for metastatic status, interdisciplinary team agreement, and hospitalization reasons. Furthermore, PALLIA-10 score independently predicted 6-month mortality, beyond other severity indicators like Karnofsky index, hospitalization reasons, tumor type, metastatic burden, team agreement, and lymphocyte count. The continuous nature of PALLIA-10 score further enhanced its prognostic value, with increasing scores correlating with reduced survival.
In our study, 82% of hospitalized cancer patients were in a palliative setting, ideally requiring palliative care. However, only 31% were receiving palliative care at enrollment, despite 70% having PALLIA-10 scores >3. While acknowledging the need for palliative care for a larger proportion, the current resources of palliative care teams are insufficient to meet this demand. Late referrals to palliative care teams, often due to reluctance to acknowledge disease progression, remain a significant barrier in comprehensive cancer centers in France. Implementing PALLIA-10 could facilitate earlier and more appropriate referrals. Simultaneously, enhancing palliative care training for oncology and nursing staff is crucial to complement specialized palliative care team interventions.
This study has limitations. It focused on cancer patients in comprehensive cancer centers, limiting generalizability to other settings. The study population was restricted to adult inpatients, excluding surgical patients and outpatients who could also benefit from early palliative care. Future research should extend to these populations and explore the role of PALLIA-10 in outpatient and early palliative care settings to improve the quality of life for a broader range of cancer patients.
Therefore, a PALLIA-10 threshold of >5, observed in over one-third of palliative patients in our study, appears more realistic and consistent with the current capacity of palliative care teams in French comprehensive cancer centers. Future randomized controlled trials are needed to evaluate the impact of PALLIA-10 implementation on palliative care referrals, patient quality of life, and caregiver burden.
Conclusion
The PALLIA-10 score, incorporating medical and psychosocial criteria, serves as a valuable decision support tool for initiating palliative care and as a prognostic indicator for overall survival. However, for hospitalized patients in comprehensive cancer centers, a threshold of >3 may be too sensitive given current palliative care resource limitations. A threshold of >5 may more effectively identify patients who urgently require palliative care team referral. Simultaneously, expanding palliative care access through outpatient and consultative models is essential for earlier intervention. Further research should focus on qualitative aspects of PALLIA-10, particularly the impact of psychosocial items, to optimize its utility as a palliative care needs assessment tool.
CONFLICT OF INTEREST
The authors declared no potential conflict of interest regarding the research, authorship, and/or publication of this article.
AUTHOR CONTRIBUTIONS
Yann MOLIN: Conceptualization, Funding acquisition, Investigation, Methodology, Writing original draft, and review; Caroline GALLAY: Investigation, Writing original draft, review; Julien GAUTIER: Conceptualization Data curation Funding acquisition Methodology Project administration resources, software, supervision, Writing ‐ original draft, and writing ‐ original draft, review; Audrey LARDY‐CLEAUD: Formal analysis, writing ‐ original draft, review; Romaine MAYET: Data curation, project administration, resources, software, supervision, writing ‐ original draft, review; Marie‐Christine GRACH: Investigation, Writing original draft, review; Gérard GUESDON: Investigation, Writing original draft, review; Géraldine CAPODANO: Investigation, Writing original draft, review; Olivier DUBROEUCQ: Investigation, Writing original draft, review; Carole BOULEUC: Investigation, Writing original draft, review; Nathalie BREMAUD: Investigation, Writing original draft, review; Anne FOGLIARINI: Investigation, Writing original draft, review; Aline HENRY: Investigation, Writing original draft, review; Nathalie CAUNES‐HILARY: Investigation, Writing original draft, review; Stéphanie VILLET: Investigation, Writing original draft, review; Christine VILLATTE Investigation, Writing original draft, review; Véronique FRASIE: Investigation, Writing original draft, review; Valérie TRIOLAIRE Investigation, Writing original draft, review; Véronique BARBAROT: Investigation, Writing original draft, review; Jean‐Marie COMMER: Investigation, Writing original draft, review; Agnès HUTIN: Investigation, Writing original draft, review; Gisèle CHVETZOFF: Conceptualization, Funding acquisition, Investigation, Methodology, supervision Writing original draft, review and editing.
All authors reviewed, discussed, and agreed to their individual contributions prior to submission.
ACKNOWLEDGMENTS
Warm thanks are firstly addressed to the patients who participated in this research. The authors wish to thank the staff in each participating site for data collection and its report in the study files (staff of palliative care teams, medical assistants, service managers, and study coordinators). Special thanks are addressed to Julie DURANTI for her help during the conception of this work and to Sophie DARNIS for medical editorial assistance with this manuscript.
Molin Y, Gallay C, Gautier J, et al. PALLIA‐10, a screening tool to identify patients needing palliative care referral in comprehensive cancer centers: A prospective multicentric study (PREPA‐10). Cancer Med. 2019;8:2950–2961. 10.1002/cam4.2118
Funding information
This research was funded by the Fondation de France, grant number 00059753 and by the Centre Léon Bérard.
REFERENCES
[References from original article – keep all of them]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Click here for additional data file. (282.6KB, pdf)
Click here for additional data file. (24.9KB, tiff)
Click here for additional data file. (29.6MB, tiff)
